Unicycive Reports Key Findings from Market Research with Nephrologists
Renazorb product profile rated as best-in-class phosphate binder
Nephrologists report high prescribing intent for Renazorb pending FDA approval
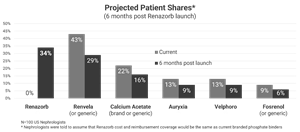
LOS ALTOS, Calif., July 20, 2022 (GLOBE NEWSWIRE) -- Unicycive Therapeutics, Inc. (Nasdaq: UNCY), a clinical stage biotechnology company developing therapies for patients with kidney disease, today announced the results from a primary market research study conducted on behalf of the company by Reason Research, LLC. The study was a discrete-choice model (“conjoint”) analysis in which 100 US-based nephrologists were presented with various phosphate binder attributes, including efficacy, safety/tolerability, pill burden, composition (calcium or non-calcium based), and dose form (chewable or swallowed), in a trade-off methodology to determine comparative prescribing preferences for currently available phosphate binders and Renazorb®, Unicycive’s product under development.
Key findings from the research:
- Nephrologists estimate that they will prescribe Renazorb (pending FDA approval) for a market-leading 34% of their dialysis patients requiring phosphate binder therapy.
- Nephrologists stated that the greatest unmet need in the treatment of hyperphosphatemia with phosphate binders is Lower pill burden/Better patient compliance
- Lower pill burden/Better patient compliance (60%), Greater efficacy (33%), Better tolerability (15%), Less frequent dosing (15%), New MOA (7%), Lower cost (5%), (unaided responses)
“This market research with nephrologists, the predominant prescribers of phosphate binders, provides strong validation for our belief in the best-in-class potential of the Renazorb product profile,” said Doug Jermasek, MBA, Executive Vice President, Corporate Strategy at Unicycive. “Nephrologists acknowledge that the greatest challenge that they face in the management of hyperphosphatemia is low patient adherence to their prescribed phosphate binders, primarily due to high pill burden. With its high phosphate binding capacity and reduced pill burden, Renazorb may offer patients a more attractive alternative to current treatment options. These survey results give us further confidence in the significant market opportunity for Renazorb. Moving forward, we are focused on establishing our global commercial strategy and launch plans for Renazorb as uncontrolled hyperphosphatemia is a persistent medical problem for CKD patients worldwide.”
The Unmet Need in Hyperphosphatemia
Renazorb is intended to be administered as a tablet to be swallowed whole at mealtimes. CKD patients typically have co-morbidities, often requiring them to be on strict pill schedules. Current phosphate binders such as, Renagel/Renvela®, Phoslo®, and Fosrenol® involve patients needing to take multiple and/or larger pills (on average, 9 pills/day), in addition to other, non-phosphate binder pills they sometimes need to take, resulting in poor adherence to the prescribed drug therapy. Potential strategies to improve adherence to phosphate binders in patients with ESRD include: (i) a reduction in pill size and number, (ii) improvement of palatability, and (iii) a reduction in associated adverse effects as published in a study by Covic and Rastogi in 2013. Consequently, Unicycive believes there is a significant need for a better phosphate binder, such as Renazorb, that has high and rapid phosphate binding, alongside a reduced pill burden for better medication compliance.
The hyperphosphatemia treatment market exceeds one billion dollars in the U.S. and is more than double that worldwide. The Unicycive team is preparing to capitalize on this substantial opportunity by offering patients and providers an attractive treatment alternative.
In tandem with the clinical development program, the Company is focused on its commercialization plans for Renazorb in the U.S. and around the world. Unicycive is conducting important market research to inform its brand and market access strategy and comprehensive launch plan for Renazorb.
About Renazorb (lanthanum dioxycarbonate)
Renazorb is a next-generation lanthanum-based phosphate binding agent utilizing proprietary nanoparticle technology being developed for the treatment of hyperphosphatemia in patients with chronic kidney disease (CKD). Its potential best-in-class profile has meaningful patient adherence benefits over currently available treatment options as it requires smaller and fewer number of pills per dose and is swallowed instead of chewed.
About Hyperphosphatemia
Hyperphosphatemia is a serious medical condition that occurs in nearly all patients with End Stage Renal Disease (ESRD). If left untreated, hyperphosphatemia leads to secondary hyperparathyroidism (SHPT), which then results in renal osteodystrophy (a condition similar to osteoporosis and associated with significant bone disease, fractures and bone pain); cardiovascular disease with associated hardening of arteries and atherosclerosis (due to deposition of excess calcium-phosphorus complexes in soft tissue). Importantly, hyperphosphatemia is independently associated with increased mortality for patients with chronic kidney disease on dialysis. Based on available clinical data to date, over 80% of patients show signs of cardiovascular calcification by the time they become dependent on dialysis.
Dialysis patients are already at an increased risk for cardiovascular disease (because of underlying diseases such as diabetes and hypertension), and hyperphosphatemia further exacerbates this. Treatment of hyperphosphatemia is aimed at lowering serum phosphate levels via two means: (1) restricting dietary phosphorus intake; and (2) using, on a daily basis, and with each meal, oral phosphate binding drugs that facilitate fecal elimination of dietary phosphate rather than its absorption from the gastrointestinal tract into the bloodstream.
Fosrenol is a Registered Trademark of Takeda Pharmaceuticals Company Ltd.
Renagel and Renvela are Registered Trademarks of Genzyme, a Sanofi Company
Phoslo and Velphoro are Registered Trademarks of Fresenius Medical Care
About Unicycive Therapeutics
Unicycive Therapeutics is a biotechnology company developing novel treatments for kidney diseases. Unicycive’s lead drug, Renazorb, is a novel phosphate binding agent being developed for the treatment of hyperphosphatemia. UNI-494 is a patent-protected new chemical entity in late preclinical development for the treatment of acute kidney injury. For more information, please visit www.unicycive.com.
Forward-looking statement
Certain statements in this press release are forward-looking within the meaning of the Private Securities Litigation Reform Act of 1995. These statements may be identified using words such as "anticipate," "believe," "forecast," "estimated" and "intend" or other similar terms or expressions that concern Unicycive's expectations, strategy, plans or intentions. These forward-looking statements are based on Unicycive's current expectations and actual results could differ materially. There are several factors that could cause actual events to differ materially from those indicated by such forward-looking statements. These factors include, but are not limited to, clinical trials involve a lengthy and expensive process with an uncertain outcome, and results of earlier studies and trials may not be predictive of future trial results; our clinical trials may be suspended or discontinued due to unexpected side effects or other safety risks that could preclude approval of our product candidates; risks related to business interruptions, including the outbreak of COVID-19 coronavirus, which could seriously harm our financial condition and increase our costs and expenses; dependence on key personnel; substantial competition; uncertainties of patent protection and litigation; dependence upon third parties; and risks related to failure to obtain FDA clearances or approvals and noncompliance with FDA regulations. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: the uncertainties related to market conditions and other factors described more fully in the section entitled ‘Risk Factors’ in Unicycive’s Annual Report on Form 10-K for the year ended December 31, 2021, and other periodic reports filed with the Securities and Exchange Commission. Any forward-looking statements contained in this press release speak only as of the date hereof, and Unicycive specifically disclaims any obligation to update any forward-looking statement, whether as a result of new information, future events or otherwise.
Investor Contact:
ir@unicycive.com
(650) 900-5470
Anne Marie
Stern Investor Relations
annemarie.fields@sternir.com
212-362-1200
SOURCE: Unicycive Therapeutics, Inc.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/c5a3d98f-a431-4283-98de-b7ad395323ae
 Source: Unicycive Therapeutics, Inc.
Source: Unicycive Therapeutics, Inc.
Released July 20, 2022